Secondary organic aerosol (SOA) formation and characterisation in AIDA
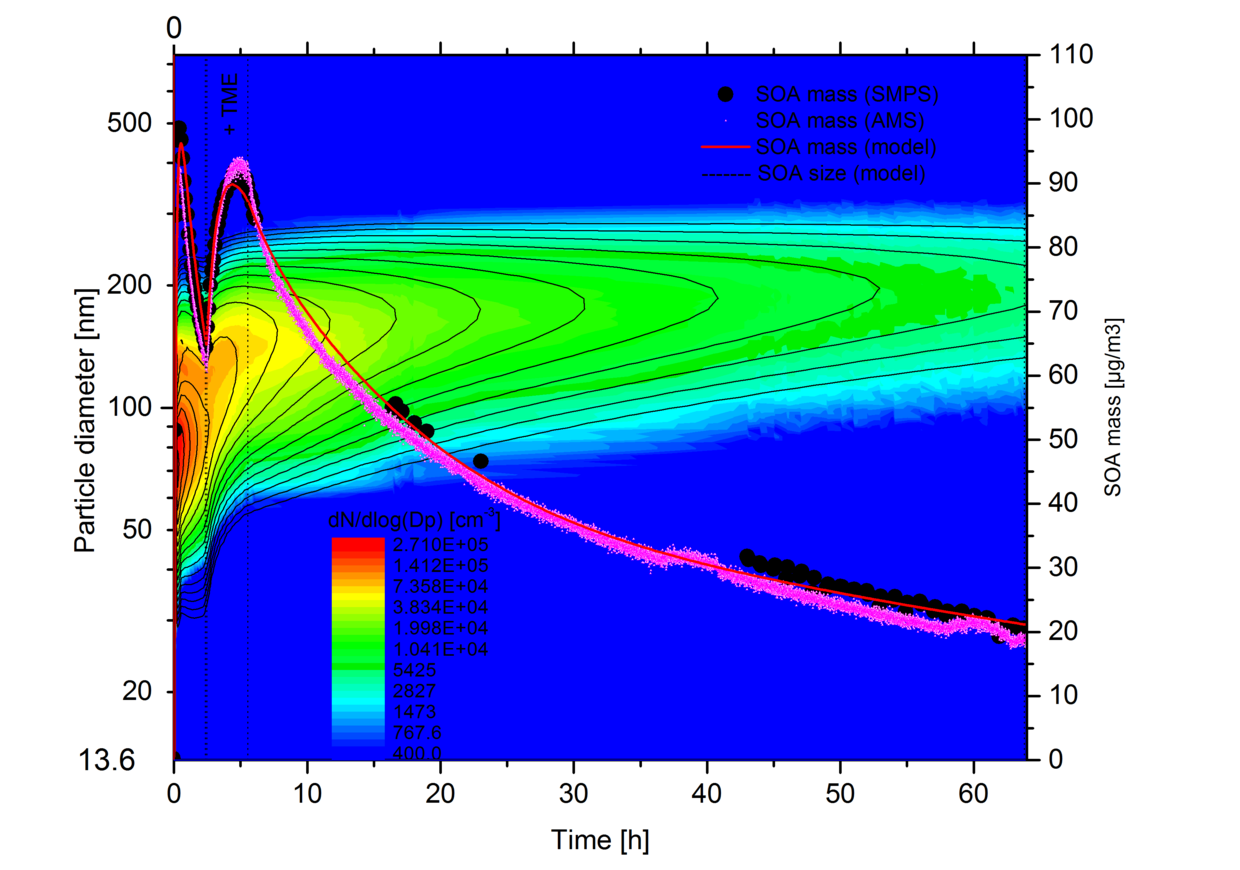
Biogenic sources dominate the global emissions of volatile organic compounds into the atmosphere. The annual emissions of biogenic volatile organic compounds (BVOC) without methane are estimated to range from 491 to 1150 Tg carbon, up to an order of magnitude more than the estimated emissions from anthropogenic sources. Almost half of the BVOC mass emitted is isoprene and about 11% are monoterpenes of which the highest fractions are α-pinene (25%) and limonene (16%). Although isoprene is the most abundant BVOC its contribution to the formation of SOA has still a high uncertainty with estimated global annual aerosol masses ranging from 10 to 120 Tg. The ozonolysis of monoterpenes is supposed to be one major source of secondary organic aerosol (SOA). The importance of monoterpene oxidation to ambient SOA is further emphasised by observations of typical oxidation products from α-pinene oxidation e.g. pinonaldehyde, pinic and pinonic acids in ambient aerosol. Global transport models predict a substantial chemical production of organic aerosol in the free troposphere and project an even higher fraction of SOA from oxidation of monoterpenes in future scenarios. However, the formulation of SOA formation and chemical composition in these models needs to be improved. Recent field measurements demonstrate that SOA is a dominant fraction of aerosol mass in the free troposphere and that theoretical estimations of SOA masses may be 1 – 2 orders of magnitude too low. Consequently, there have been a number of laboratory studies focusing on SOA yields, i.e. the mass of SOA produced per mass of reacted precursor. These depend mainly on the organic precursor molecules and on the oxidants initiating the degradation (OH, O3, NO3). However, it has also been shown that other factors influence the SOA yield, i.e. the mass of organic aerosol that can serve as solvent for semivolatile organic compounds, the acidity of and chemical reactions in the particulate phase, the humidity, and the temperature that controls the phase equilibrium of the semivolatile organics and also influences the reaction pathways leading to condensable molecules. There has been a considerable amount of work on aerosol yields from the ozonolysis of α-pinene or limonene near room temperature (290-303 K). Studies covering more extended temperature ranges are less abundant. Obviously, temperature dependences are important in describing SOA formation under tropospheric conditions and – when properly evaluated - can improve our understanding of the physical and chemical mechanisms involved in the ozonolysis of monoterpenes. Experiments in large environmental chambers covering a large temperature range are particularly suited to derive parameterisations which are valid under real world conditions.
- Yields of SOA from different precursors (e.g. pinene, limonene, isoprene) as function of temperature, humidity, radical scavenger
- Role of oxidising agent (e.g. ozone, OH radicals)
- Partitioning of semi volatile reaction products
- Reaction mechanisms for SOA formation and ageing
- Kinetics of gas phase and heterogeneous reactions
- Changing particle volatility
- Photochemistry
- Optical properties of SOA
- Glass transitions of SOA
- Ice nucleation properties of SOA (see cloud microphysics)
- Impact of SOA coatings on physical properties of other aerosol particles
For further information please contact Dr. Harald Saathoff.
References:
- Seasonal characteristics of organic aerosol chemical composition and volatility in Stuttgart, Germany
Huang W., Saathoff H., Shen X., Ramisetty R., Leisner T., and Mohr C.,
Atmos. Chem. Phys. 9, 19, 11687-11700, https://doi.org/10.5194/acp-19-11687-2019 (2019) - Chemical Characterization of Highly Functionalized Organonitrates Contributing to Night-time Organic Aerosol Mass Loadings and Particle Growth
Huang, W., Saathoff, H., Shen, X., Ramisetty, R., Leisner, T., and Mohr C.,
Environ. Sci. Technol. 9, 53, 1165-1174, DOI: 10.1021/acs.est.8b05826 (2019) - α-pinene secondary organic aerosol at low temperature: Chemical composition and implications for particle viscosity
Huang, W., Saathoff, H., Pajunoja, A., Shen, X., Naumann, K.-H., Wagner, R., Virtanen, A., Leisner, T., Mohr, C.,
Atmos. Chem. Phys. 9, 18, 2883-2898, doi.org/10.5194/acp-18-2883-2018, (2018) - Aging of biogenic secondary organic aerosol via gas-phase OH radical reactions
N. Donahue, K. M. Henry, T. F. Mentel, A. Kiendler-Scharr, C. Spindler, B. Bohn, T. Brauers, H. P. Dorn, H. Fuchs, R. Tillmann, A. Wahner, H. Saathoff, K.-H. Naumann, O. Möhler, T. Leisner, L. Müller, M.-C. Reinnig, T. Hoffmann, K. Salo, M. Hallquist, M. Frosch, M. Bilde, T. Tritscher, P. Barmet, A. P. Praplan, P. F. DeCarlo, J. Dommen, A.S.H. Prévôt, and U. Baltensperger
Proc. Natl. Acad. Sci. USA 109 (34), 13503-13508 (2012) - Influence of relative humidity and temperature on the production of pinonaldehyde and OH radicals from the ozonolysis of α-pinene
R. Tillmann, M. Hallquist, Å. M. Jonsson, A. Kiendler-Scharr, H. Saathoff, Y. Iinuma, and Th. F. Mentel
Atmos. Chem. Phys. 10, 7057-7072 (2010) - Temperature dependence of yields of secondary organic aerosols from the ozonolysis of α-pinene and limonene
H. Saathoff, K.-H. Naumann, O. Möhler, Å. M. Jonsson, M. Hallquist, A. Kiendler-Scharr, T. Mentel, R. Tillmann, and U. Schurath
Atmos. Chem. Phys. 9, 1551 (2009) - Temperature dependence of the rate coefficient for the α-pinene reaction with ozone in the range between 243 K and 303 K
R. Tillmann, H. Saathoff, T. Brauers, A. Kiendler-Scharr, and T. F. Mentel
Phys. Chem. Chem. Phys. 11, 2323 (2009) -
The effect of organic coating on the heterogeneous ice nucleation efficiency of mineral dust aerosols
O. Möhler, S. Benz, H. Saathoff, M. Schnaiter, R. Wagner, J. Schneider, S. Walter, V. Ebert, and S. Wagner
Environ. Res. Lett. 3, 025007 (2008) -
Volatility of secondary organic aerosols from the ozone initiated oxidation of α-pinene and limonene
Å. M. Jonsson, M. Hallquist, and H. Saathoff
J. Aerosol Sci. 38, 843, (2007)
